Classify Elements Activity: 8 Hands-On Stations for Teaching Metals, Nonmetals, and Metalloids on the Periodic Table (TEKS 6.6C)
Drop a wall-sized periodic table on the projector and ask a 6th grader, "Why are some elements yellow on this map and other elements blue?" Most can't tell you. They've seen the periodic table since 4th grade, but the colored sections are just decoration to them. They don't know that the colors mean something. They don't know that you can predict whether a substance is shiny or dull, conducts electricity or insulates against it, just by where it sits on the table.
TEKS 6.6C is where that finally clicks. The standard asks students to classify elements as metals, nonmetals, or metalloids using their physical properties on the periodic table. It's the first time most kids learn that the table isn't just a list of names. It's a tool that groups elements by behavior.
The Classify Elements Station Lab for TEKS 6.6C closes that gap in one to two class periods. Kids run real samples through three property tests (appearance, conductivity with a tester, malleable vs. brittle with a small hammer), examine reference cards comparing metals, nonmetals, and metalloids on heat resistance, conductivity, luster, and density, and finish by coloring a periodic table to show the staircase line that splits metals from nonmetals. By the end, they can pick up an element sample, predict its category, and explain why.
8 hands-on stations for teaching how to classify elements
A station lab is a student-led activity where small groups rotate through 8 stations (plus a 9th challenge station for early finishers) at their own pace during one to two class periods. You become a facilitator instead of a lecturer. You walk around, spot-check, and break misconceptions while kids work through the rotation.
The Classify Elements Station Lab has four input stations (where students take in new info on metals, nonmetals, metalloids, and the periodic table) and four output stations (where they show what they learned). Here's what's at each one.
4 input stations: how students learn to classify elements
A short YouTube video introduces how scientists classify elements based on their physical and chemical properties. Kids answer three questions: how elements are classified, examples of metal properties from the video, and what makes metalloids different from metals and nonmetals. Visual learners hook in fast at this station because the video shows actual samples and where they land on the periodic table, which is hard to picture from words alone.
A one-page passage called "The Building Blocks of Everything!" frames the periodic table as a mosaic. Each tile is an atom, and elements are grouped into families based on shared properties. The passage walks through metals (luster, strength, conductivity, found mostly on the left), nonmetals (dull, poor conductors, on the right), metalloids (silicon, arsenic, used in semiconductors for phones and computers), and rare earth elements (scarce metals used in magnets and electronics). Vocabulary is bolded throughout. Three multiple-choice questions follow, plus the vocab notes section. Comes in two reading levels (Dependent and Modified) plus a Spanish version.
This is the heart of the lab. Three-test physical-properties investigation. Test 1 (Appearance): kids place small samples on a paper towel and record color, luster, and form (sheet, powder, crystal, gas). Test 2 (Conductivity): they touch a battery-powered conductivity tester to each sample. If the bulb lights, it's a conductor. If not, an insulator. They predict what a semi-conductor would do. Test 3 (Malleable vs. Brittle): kids try to bend each sample with their fingers, then tap gently with a small hammer. Malleable flattens. Brittle shatters. After all three tests, they classify each sample as metal, nonmetal, or metalloid and justify their answer with the test data.
Students examine 8 reference cards: real-life examples of metals (Fe, Cu, Au, Pt, Ti, Sn, Co, Ni, Pb, Ag, Zn), examples of nonmetals (gas tanks of nitrogen, oxygen, helium, argon), examples of metalloids (a chunk of silicon), and a full periodic table. The next four cards define each property (heat resistance, conductivity, malleability, luster, density, state of matter) and end with a comparison chart showing how metals, nonmetals, and metalloids stack up on every property. Six questions follow, including a mystery-element challenge: kids get a list of properties (gray, solid at room temperature, semiconducting, density 2.33 g/cm³) and have to classify it as metal, nonmetal, or metalloid using reasoning.

4 output stations: how students show what they learned
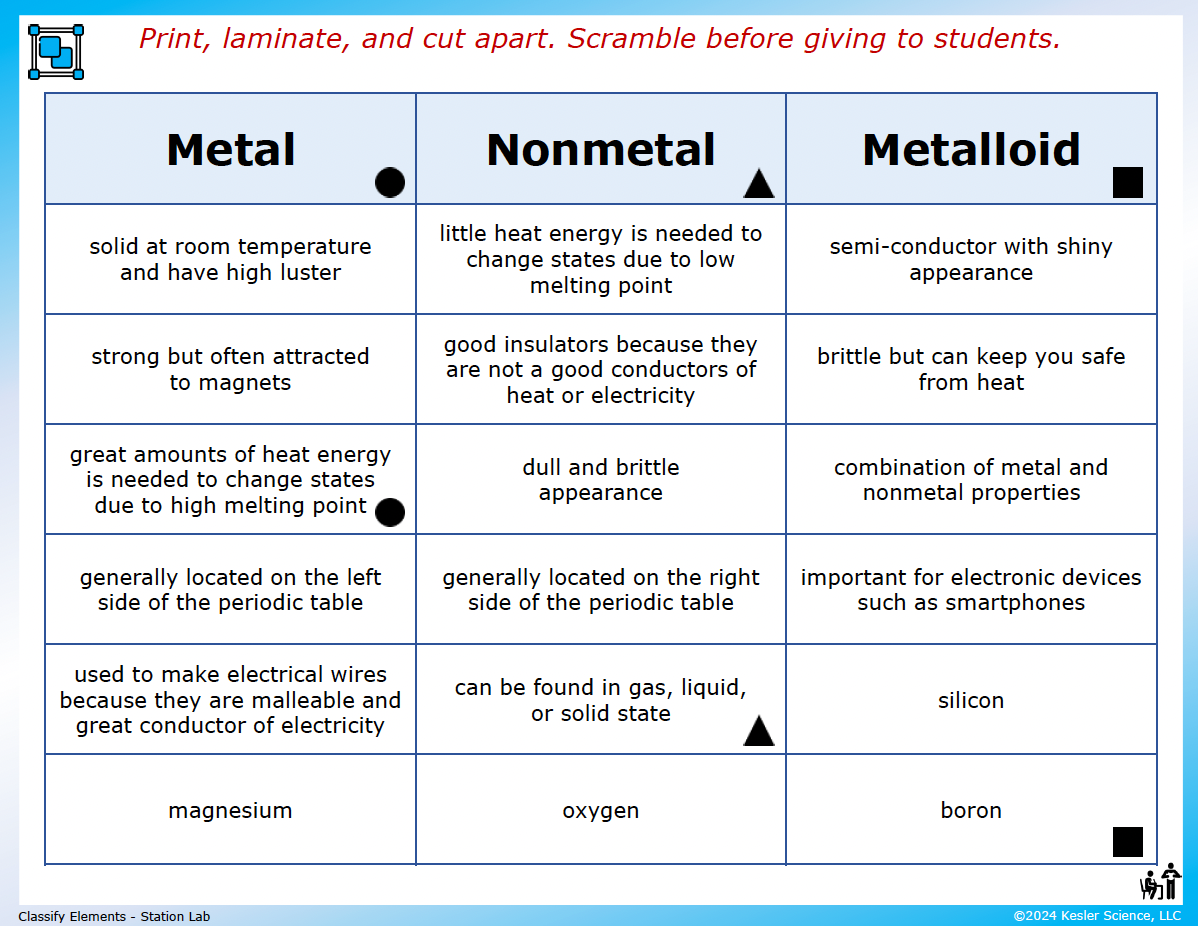
A three-column card sort. Kids sort 18 description cards into METAL, NONMETAL, and METALLOID columns. Cards include "solid at room temperature and have high luster," "good insulators because they are not good conductors of heat or electricity," "semi-conductor with shiny appearance," "used to make electrical wires because they are malleable," "can be found in gas, liquid, or solid state," plus a representative element for each column (magnesium for metal, oxygen for nonmetal, boron for metalloid, plus silicon). Easy to spot-check at a glance.
Students color a printed periodic table to show how the elements are categorized. They trace the zigzag staircase line in black from boron through polonium that separates metals from nonmetals. Then they color nonmetals blue (H, He, C, N, O, F, P, S, Cl, Ar, Br, Kr, etc.), metalloids green (B, Si, Ge, As, Sb, Te, Po), all metals yellow, and rare earth elements orange (the two rows starting with La and Ac). Even kids who say "I can't draw" surprise themselves here because the geography of the table is itself the diagram.
Three open-ended questions: why scientists need a way to group elements together, the differences between metals and nonmetals, and what rare earth metals are and how we use them in daily life (magnets, catalysts, electronic devices). Forces kids to commit definitions to paper and connect classification to real-world technology. This is the writing practice middle schoolers need and rarely get in science class.
Eight multiple-choice and fill-in-the-paragraph questions tied to TEKS 6.6C vocabulary (metal, nonmetal, metalloid, periodic table, rare earth element). Includes which is the largest group on the periodic table, why rare earth elements matter for modern tech, and how metalloids combine traits of both metals and nonmetals. The fill-in paragraph weaves all five vocabulary words together into a single explanation. If you're grading the lab, this is the easiest station to grade.
Bonus Challenge It! station for early finishers
Four optional extensions: write a Rental Space ad for a family of the periodic table (list desired characteristics for becoming a resident), research the differences between 10K, 14K, 18K, and 24K gold, design a custom periodic table for a non-science theme (superpowers, foods, sports) with 20 items organized by shared properties, or create Mystery Pixel Art where players color element boxes based on clues to reveal a hidden picture. Requires teacher approval before they start.
How this fits into a complete classify elements unit
This Station Lab is the Explore day of our full Classify Elements Complete 5E Lesson for TEKS 6.6C. The complete two-week unit follows the 5E method of instruction and includes an Engage hook, the Classify Elements Station Lab for Explore, PowerPoint slides and interactive notebook pages for Explain, student choice projects to Elaborate, and an Evaluate assessment.
Most teachers grab the full 5E because the Station Lab lands hardest with the days around it. But if you just need a strong hands-on day on metals, nonmetals, and metalloids, the Station Lab on its own does the job.
Materials needed to teach how to classify elements
Materials beyond what's in the download:
- A small set of labeled element samples per station. A typical chemistry-class set works (a strip of magnesium, a piece of copper wire, sulfur powder, carbon (graphite or pencil lead), aluminum foil, silicon wafer if you have one). Three or four samples is plenty.
- One battery-powered conductivity tester per station rotation. A simple kit with a battery, two wires, and a small bulb works. Many teachers DIY one with a 9V battery, alligator clips, and an LED.
- A small hammer or rubber mallet for the malleable-vs.-brittle test. Set the sample on a piece of paper on the floor or a sturdy lab table.
- A wall-sized or printed periodic table for reference during Explore It! and Research It!.
- Colored pencils or markers (yellow, blue, green, orange, black) for the Illustrate It! station's periodic-table coloring.
- Pencils and the printed answer sheets (included)
- A device with internet for the Watch It! station
Standard covered: Texas TEKS 6.6C —
Classify substances by their physical properties using the periodic table. Supporting Standard.
See the full standard breakdown →Grade level: 6th grade physical science
Time: One to two class periods (45–110 minutes total). Plan for two periods the first time you run a station lab.
Common student misconceptions this lab fixes
- "Metals are anything that looks shiny. If it's dull, it's a nonmetal."
This is the surface-level rule kids invent on day one, and it falls apart fast. The Explore It! conductivity test catches it first. Some metals (like iron filings) look dull because of oxidation, but they still light up the bulb on the conductivity tester. The Research It! comparison chart spells it out: luster is just one property. Heat resistance, conductivity, malleability, density, and state of matter all matter. The mystery-element challenge on Research It! Card 12 forces kids to use multiple properties at once (boiling point, density, semiconducting behavior) to classify an element instead of relying on appearance alone. By the end, they're using a property checklist instead of a vibe check.
- "Metalloids are just metals that aren't very good. They're broken metals."
Many 6th graders treat metalloids as a leftover category. The Read It! passage corrects this directly: metalloids combine the best of both worlds, which is exactly why we use them in semiconductors. Silicon isn't a failed metal. It's the foundation of every smartphone, computer chip, and solar panel because it sits between metal and nonmetal behavior on conductivity. The Research It! comparison chart shows metalloids as their own column with "semi-conductor of heat" and "semi-conductor of electricity" and "varies based on element." The Organize It! card sort then forces kids to put "important for electronic devices such as smartphones" in the metalloid column. Once they connect metalloids to the phone in their pocket, the category sticks.
- "The periodic table is just a list of element names. The colors and groups don't really mean anything."
This one is everywhere. Kids treat the periodic table like a phone book. They use it to look up symbols and atomic numbers but don't realize the layout itself encodes information. The Illustrate It! station fixes this directly. By coloring the table (metals yellow, nonmetals blue, metalloids green, rare earth elements orange) and tracing the staircase line from boron through polonium, kids physically experience that position predicts properties. An element on the bottom-left will be a metal, no matter what its name is. An element on the top-right will be a nonmetal. The Research It! Card 4 makes the same point. The table isn't a list. It's a map.
What you get with this classify elements activity
When you buy the Station Lab, you get a single download with everything you need:
- Print version at two reading levels (Dependent for on-grade, Modified for additional support) plus a Spanish Read It! passage
- Digital version as PowerPoint files (works in Google Slides too) at both levels for 1:1 classrooms or Google Classroom
- Teacher Directions and Answer Key for both versions, all keys included
- Station task cards ready to print, laminate, and drop in baskets at each station
- Reference cards for the Research It! station (examples of metals/nonmetals/metalloids, full periodic table, six property cards, and the metals-vs.-nonmetals-vs.-metalloids comparison chart)
- Sort cards for the Organize It! station (18 description cards plus three header cards)
- Blank periodic table for the Illustrate It! station coloring activity
- Student answer sheets for each level
Tips for teaching how to classify elements in your 6th grade classroom
Two things make this lab go smoother the first time:
1. Test the conductivity tester before class.
The Explore It! station works because kids see the bulb light up for metals and stay dark for nonmetals. The first time I ran a similar setup I didn't check the battery, and the bulb wouldn't light for anything. Three groups rotated through and decided everything was a nonmetal. Set up one tester before class, touch it to a known conductor (a paperclip or a piece of metal foil), and confirm the bulb lights. Bring a spare battery and a backup LED in case mid-rotation gear fails.
2. Skip the hammer for soft samples.
The malleable-vs.-brittle test calls for tapping samples with a small hammer. For soft metals like aluminum foil, a hammer is overkill (they bend with finger pressure). For brittle samples like sulfur powder or carbon graphite, a hammer just makes a mess. I tell groups to test bendiness with their fingers first. If it bends, it's malleable. Done. Save the hammer for samples that resist finger pressure (a copper wire or a magnesium strip). It cuts the cleanup time in half and keeps the focus on the result, not the tool.
Get this classify elements activity
Or if you want the full two-week experience with the Engage hook, Explain day, Elaborate extension, and Evaluate assessment all included:
(Station Lab is included)
Frequently asked questions
What does TEKS 6.6C cover?
Texas TEKS 6.6C asks 6th grade students to classify substances by their physical properties using the periodic table. Students should be able to identify an unknown element as a metal, nonmetal, or metalloid based on properties like luster, conductivity, malleability, and density, and explain how the periodic table organizes elements into families with shared behavior. The standard pairs naturally with 6.6A (particles and kinetic energy) and 6.6B (pure substances and mixtures).
Is this kids' first time with the periodic table?
Most 6th graders have seen a periodic table before, but this is often the first time they're asked to use it as a tool. The Read It! passage introduces the table as a map of element families, the Research It! card stack drives home that location predicts properties, and the Illustrate It! coloring activity locks in where metals, nonmetals, metalloids, and rare earth elements actually live. By the end, the table stops being a wall of names and becomes a usable reference.
How long does this classify elements activity take?
One to two class periods (45 to 110 minutes total). The Explore It! station's three property tests (appearance, conductivity, malleable vs. brittle) are the longest piece, so plan for two periods the first time you run a station lab. Once your class has the rotation routine down, most groups can finish all 8 stations in one period.
Do I need element samples for this?
Helps a lot but isn't required. A typical chemistry classroom kit (a strip of magnesium, copper wire, sulfur, carbon, aluminum foil) works perfectly. If you don't have samples, kids can run the Explore It! tests on common classroom objects (a paperclip for iron, pencil lead for carbon, foil for aluminum) and get most of the same results. Total cost for a full element kit: under $30 from a science-supply vendor, or you can borrow one from a high school chemistry teacher.
Can I use this in a 1:1 digital classroom?
Yes. The full digital version (PowerPoint or Google Slides) works in 1:1 classrooms and Google Classroom. Students drag digital cards for the Organize It! sort and color a digital periodic table for Illustrate It!. The Explore It! hands-on tests are harder to digitize, but you can substitute conductivity simulation videos or PhET particle simulations if you don't have the supplies.
Related resources
- Texas teacher? See the full TEKS 6.6C standard breakdown for misconceptions, phenomena, and engagement ideas.
- Teaching 6.6B first? Check out our Pure Substances and Mixtures Station Lab for TEKS 6.6B, where students learn to classify mixtures as homogeneous or heterogeneous before sorting elements.
- Heading into 6.6D next? See our Comparing Density Station Lab for TEKS 6.6D, where students measure and compare the density of liquids and solids.


