Pure Substances and Mixtures Activity: 8 Hands-On Stations for Teaching Mixtures, Solutions, and Heterogeneous vs. Homogeneous (TEKS 6.6B)
Hold up a glass of saltwater in front of a 6th grader and ask, "Is this one thing or two things?" You'll get a split room. Some kids see clear liquid and call it one substance. Others remember the salt got stirred in and call it two. Then ask the follow-up: "Could you get the salt back if you wanted to?" That question is where pure substances and mixtures actually click.
TEKS 6.6B is where 6th graders meet this idea for the first time. They have to compare and contrast pure substances (like pure water or iron) with mixtures (like saltwater, smog, or pizza), and they have to recognize the special kind of mixture called a solution. It sounds simple until they try to sort lemonade, oil-and-water, and a chocolate chip cookie into the right buckets.
The Pure Substances and Mixtures Station Lab for TEKS 6.6B closes the gap in one to two class periods. Kids dissolve different amounts of salt into water to compare two solutions side by side, scoop random colored beads to model a heterogeneous mixture, examine reference cards showing pure substances vs. homogeneous and heterogeneous mixtures, and finish by sorting 15 example items (oxygen, glue, smog, pizza, lemonade) into the correct categories. By the end, they can look at any sample and tell you whether it's pure, homogeneous, heterogeneous, or a solution.
8 hands-on stations for teaching pure substances and mixtures
A station lab is a student-led activity where small groups rotate through 8 stations (plus a 9th challenge station for early finishers) at their own pace during one to two class periods. You become a facilitator instead of a lecturer. You walk around, spot-check, and break misconceptions while kids work through the rotation.
The Pure Substances and Mixtures Station Lab has four input stations (where students take in new info on pure substances, mixtures, and solutions) and four output stations (where they show what they learned). Here's what's at each one.

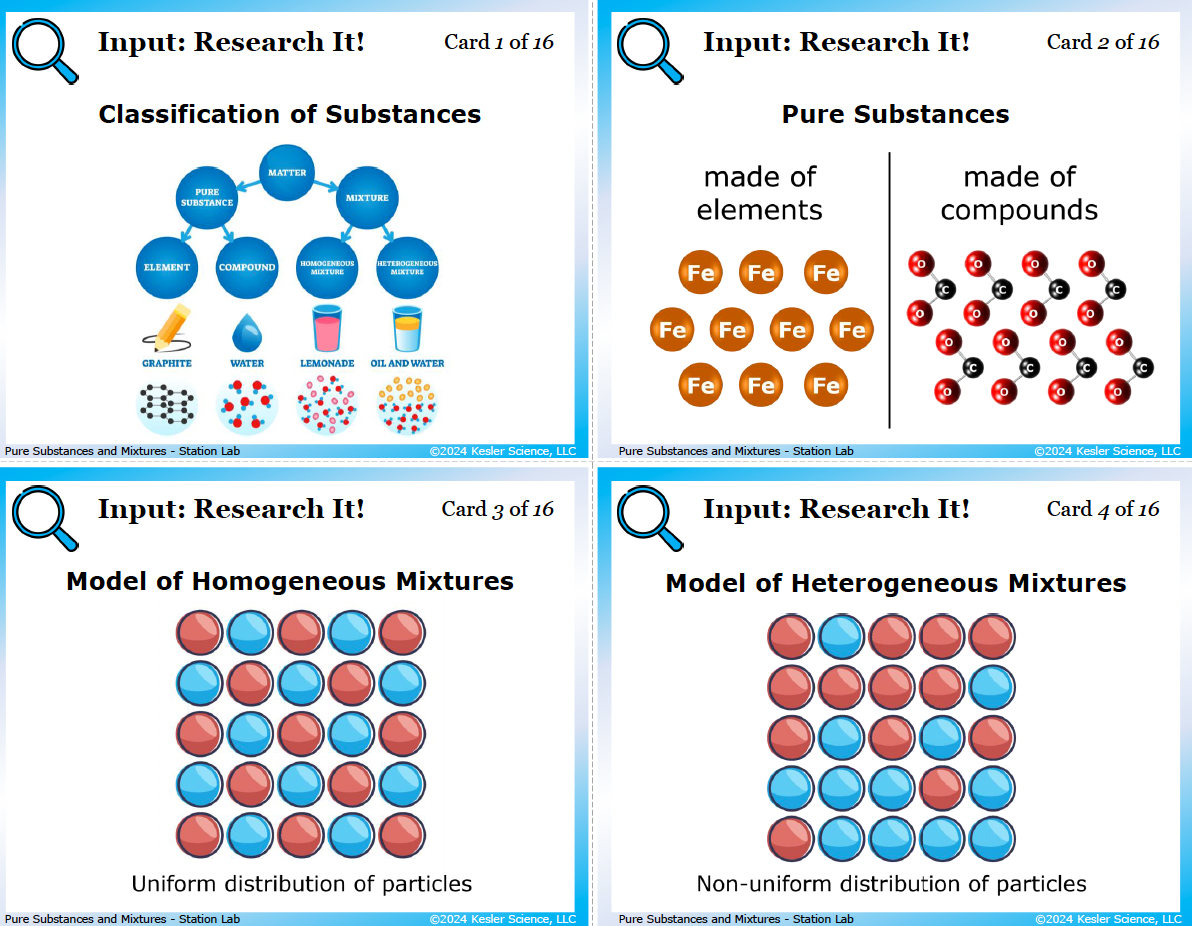
4 input stations: how students learn pure substances and mixtures
A short YouTube video introduces matter and walks kids through the difference between pure substances and mixtures, plus the two types of mixtures (homogeneous and heterogeneous). Kids answer three quick questions: how all matter on Earth can be classified, an example of a pure substance, and the two types of mixtures. Visual learners hook in fast at this station because the video shows particle-level differences they can't picture from words alone.
A one-page passage called "A Mixture of Smog" frames the lesson around pollution hanging over a city. The passage walks through pure substances (always identical samples), mixtures (components keep their own properties and can be physically separated), homogeneous mixtures (evenly distributed), heterogeneous mixtures (uneven, like sand in water), and solutions (homogeneous mixtures where one substance dissolves into another). Glue, saltwater, and smog all show up as concrete examples. Vocabulary is bolded throughout. Three multiple-choice questions follow, plus the vocab notes section. Comes in two reading levels (Dependent and Modified) plus a Spanish version.
This is the heart of the lab. Two hands-on activities. Activity 1: Kids make two saltwater solutions, one with 1 g of salt in 10 mL of water and one with 3 g of salt in 10 mL of water. They stir until the salt dissolves, then answer whether saltwater is a pure substance and whether it qualifies as a solution. Activity 2: Kids scatter mixed-color beads on the table, close their eyes, scoop 25 random beads into a cup, and explain why their bead cup is a heterogeneous mixture and how they could turn it into a homogeneous one. Plus four definition cards on the table (pure substance, mixture, homogeneous, heterogeneous) for kids who blank on the names.
Students examine 10 reference cards: a Classification of Substances tree (matter splits into pure substance and mixture, then into element/compound and homogeneous/heterogeneous, with graphite, water, lemonade, and oil-and-water as examples), particle-level models of pure substances and both types of mixtures, real-life examples (steel, air, rain, lemonade for homogeneous; ice in soda, soil, cereal in milk, oil and vinegar for heterogeneous), and a card explaining that solutions are homogeneous mixtures where something dissolves. Six questions follow, including one that uses a pizza and asks kids to identify the components and classify the type of mixture.

4 output stations: how students show what they learned
A three-column card sort. Kids sort 15 example cards into PURE SUBSTANCES, HOMOGENEOUS MIXTURES, and HETEROGENEOUS MIXTURES. Pure substances: oxygen, iron, ice, salt, gold. Homogeneous: air, salt water, glue, milk, lemonade. Heterogeneous: chocolate chip cookie, pizza, chicken noodle soup, smog, soil. The pure-substance side gets a periodic-table icon next to each example, which reinforces the element/compound idea from the Research It! card. Easy to spot-check at a glance.
Students draw four boxes side by side, labeled pure substance, homogeneous mixture, heterogeneous mixture, and solution. Inside each box they sketch images or particle diagrams that show the physical properties of that type and label every part. The drawing forces them to remember that homogeneous and solutions look uniform at the particle level while heterogeneous shows clumps and uneven distribution. Even kids who say "I can't draw" surprise themselves here.
Three open-ended questions: define a heterogeneous mixture and give an example, define a homogeneous mixture and give an example, and list five examples of pure substances. Forces kids to commit to definitions in their own words and pull examples from memory. This is the writing practice middle schoolers need and rarely get in science class.
Eight multiple-choice and fill-in-the-paragraph questions tied to TEKS 6.6B vocabulary (pure substance, mixture, homogeneous mixture, heterogeneous mixture, solution). Includes how to identify a heterogeneous mixture, a definition question on solutions, and which of four options (chocolate milk, oxygen, trail mix, pizza) is the pure substance. The fill-in paragraph weaves all five vocabulary words together into a single explanation. If you're grading the lab, this is the easiest station to grade.
Bonus Challenge It! station for early finishers
Four optional extensions: research a real-world homogeneous mixture and explain how it affects daily life, design a public-awareness campaign poster about the components of smog (and classify each as a pure substance, homogeneous mixture, or heterogeneous mixture), create an original infographic explaining all three categories, or write a song about pure substances and mixtures. Requires teacher approval before they start.
How this fits into a complete pure substances and mixtures unit
This Station Lab is the Explore day of our full Pure Substances and Mixtures Complete 5E Lesson for TEKS 6.6B. The complete two-week unit follows the 5E method of instruction and includes an Engage hook, the Pure Substances and Mixtures Station Lab for Explore, PowerPoint slides and interactive notebook pages for Explain, student choice projects to Elaborate, and an Evaluate assessment.
Most teachers grab the full 5E because the Station Lab lands hardest with the days around it. But if you just need a strong hands-on day on pure substances, mixtures, and solutions, the Station Lab on its own does the job.
Materials needed to teach pure substances and mixtures
Materials beyond what's in the download:
- Two beakers (or clear cups) per station rotation for the Explore It! Activity 1 saltwater solutions.
- Distilled water (about 20 mL per group) and table salt (4 g per group) for the saltwater activity.
- A stirring rod or plastic spoon for stirring the saltwater.
- A small cup and a handful of mixed-color beads for the Explore It! Activity 2 (pony beads work great). Pre-mix 4 to 5 colors so the random scoop produces a clearly heterogeneous mix.
- Colored pencils or markers for the Illustrate It! station.
- Pencils and the printed answer sheets (included)
- A device with internet for the Watch It! station
Standard covered: Texas TEKS 6.6B —
Compare and contrast a variety of mixtures, including solutions. Supporting Standard.
See the full standard breakdown →Grade level: 6th grade physical science
Time: One to two class periods (45–110 minutes total). Plan for two periods the first time you run a station lab.
Common student misconceptions this lab fixes
- "If I dissolve salt in water, it disappears. So saltwater is just water now."
This is the big one. Sixth graders see salt vanish into water and assume it stopped existing. The Explore It! Activity 1 fixes it directly. They make two saltwater solutions with different amounts of salt (1 g vs. 3 g in the same 10 mL of water) and taste or compare them. The 3-gram cup is noticeably saltier. The salt is still there, just spread evenly throughout the water at the particle level. The Read It! passage reinforces this with the line that water can evaporate and leave the salt behind. Once kids realize the salt is hiding, not gone, the idea that solutions are mixtures (not pure substances) finally lands.
- "Pure means clean. Tap water is pure water."
Lots of 6th graders confuse "pure" with "clean" or "safe to drink." In chemistry, pure means every sample is identical at the particle level. Tap water is a mixture (water plus minerals plus chlorine plus whatever else is in your pipes). The Read It! passage spells it out: pure water is always the same, but glue is a homogeneous mixture because different batches have different ingredients in different amounts. The Research It! Pure Substances card shows iron atoms (an element) and a compound, both with the same arrangement repeated identically. The Organize It! card sort then forces them to put oxygen, iron, ice, salt, and gold in the pure pile and milk, lemonade, and air in the homogeneous mixture pile. The categorization fights the everyday meaning.
- "If a mixture looks the same all the way through, it must be a pure substance."
This is the trickiest one for 6th graders, especially with liquids. Lemonade looks uniform. Air looks like nothing. Salt water looks like clear water. Kids think "if I can't see the parts, it's pure." The Research It! reference cards correct this by showing the particle-level model: homogeneous mixtures have two different colored spheres distributed evenly, but they are still two separate substances. Lemonade has water plus sugar plus lemon juice, all evenly mixed. The fact that you can't see the parts doesn't mean they aren't there. The Organize It! sort drives it home with milk, glue, salt water, and air all sitting in the homogeneous mixture column instead of pure.
What you get with this pure substances and mixtures activity
When you buy the Station Lab, you get a single download with everything you need:
- Print version at two reading levels (Dependent for on-grade, Modified for additional support) plus a Spanish Read It! passage
- Digital version as PowerPoint files (works in Google Slides too) at both levels for 1:1 classrooms or Google Classroom
- Teacher Directions and Answer Key for both versions, all keys included
- Station task cards ready to print, laminate, and drop in baskets at each station
- Reference cards for the Research It! station (classification tree, particle-level models of pure substances and mixtures, real-life examples for both types of mixtures)
- Sort cards for the Organize It! station (15 example cards plus three header cards for pure substances, homogeneous, and heterogeneous mixtures)
- Definition cards for the Explore It! station (pure substance, mixture, homogeneous, heterogeneous)
- Student answer sheets for each level
Tips for teaching pure substances and mixtures in your 6th grade classroom
Two things make this lab go smoother the first time:
1. Pre-measure the salt and water for the Explore It! station.
Activity 1 has groups making two saltwater solutions with specific amounts (1 g salt in 10 mL water and 3 g salt in 10 mL water). The first time I ran a similar setup, kids spent ten minutes wrestling with the digital scale and graduated cylinders. Now I pre-portion the salt into small cups (one labeled 1g, one labeled 3g) and the water into pre-marked beakers. The activity then takes three minutes instead of fifteen, which keeps the rotation moving.
2. Mix at least 4 colors of beads for Activity 2.
The Explore It! Activity 2 only works if the random scoop is visibly heterogeneous. If you only mix two colors of beads, kids might pull a cup that's mostly one color and miss the point. Mixing four or five colors of pony beads guarantees that every random 25-bead scoop looks clearly uneven, which makes the heterogeneous label obvious. It also sets up the follow-up question (how would you make this a homogeneous mixture?) since the answer is to scoop equal numbers of each color in a repeating pattern.
Get this pure substances and mixtures activity
Or if you want the full two-week experience with the Engage hook, Explain day, Elaborate extension, and Evaluate assessment all included:
(Station Lab is included)
Frequently asked questions
What does TEKS 6.6B cover?
Texas TEKS 6.6B asks 6th grade students to compare and contrast a variety of mixtures, including solutions. Students should be able to look at any sample of matter and tell you whether it's a pure substance, a homogeneous mixture, a heterogeneous mixture, or a solution, and explain why. The standard is part of the 6.6 matter and energy strand and pairs naturally with 6.6A (particles and kinetic energy) and 6.6C (classifying elements on the periodic table).
Is this kids' first time meeting solutions and mixtures?
Yes for most 6th graders. They've seen mixtures in earlier grades (trail mix, sand in water) but "homogeneous" and "heterogeneous" as formal categories are brand new. The Read It! smog passage introduces both in bold, the Research It! reference cards show the particle-level difference, and the Organize It! sort tests whether they can apply the categories to 15 real-world examples.
How long does this pure substances and mixtures activity take?
One to two class periods (45 to 110 minutes total). The Explore It! station's two activities (saltwater solutions and the bead scoop) are the longest pieces, so plan for two periods the first time you run a station lab. Once your class has the rotation routine down, most groups can finish all 8 stations in one period.
Do I need a lot of supplies for this?
Pretty light. Two clear cups or beakers per station, a small amount of distilled water and table salt, a stirring rod, and a cup of mixed-color pony beads. Total cost for a class of 30: under $10 if you're starting from nothing. The Watch It! station also needs a device with internet.
Can I use this in a 1:1 digital classroom?
Yes. The full digital version (PowerPoint or Google Slides) works in 1:1 classrooms and Google Classroom. Students drag digital cards for the Organize It! sort instead of physically arranging them. The Explore It! hands-on activities (saltwater solutions and the bead scoop) are harder to digitize, but you can substitute simulation videos or particle-level animations if you don't have the supplies.
Related resources
- Texas teacher? See the full TEKS 6.6B standard breakdown for misconceptions, phenomena, and engagement ideas.
- Teaching 6.6A first? Check out our Matter and Kinetic Energy Station Lab for TEKS 6.6A, where students learn how particles in solids, liquids, and gases differ in structure and motion.
- Heading into 6.6C next? See our Classify Elements Station Lab for TEKS 6.6C, where students sort metals, nonmetals, and metalloids on the periodic table.


