Comparing Density Activity: 8 Hands-On Stations for Teaching Density of Liquids and Solids (TEKS 6.6D)
Hand a 6th grader a small wooden block and a much larger Styrofoam ball, then ask, "Which one is heavier?" Most of them will pick the bigger one without thinking. Then drop both in a tub of water and watch their faces. The wood block sinks. The Styrofoam ball floats. Now they have to explain why bigger isn't the same as heavier, and why a heavy ship made of steel still floats. That's density.
TEKS 6.6D asks 6th graders to calculate density of solids and liquids and to predict whether a substance will sink or float in another substance based on its density. They have to learn the density formula (D = m/V), recognize the units (g/cm³ for solids, g/mL for liquids and gases), and use density values to explain why a lava lamp blob rises and falls.
The Comparing Density Station Lab for TEKS 6.6D closes that gap in one to two class periods. Kids build a real density column with corn syrup, water, and vegetable oil, then drop in a coin, a glass marble, a cork, and a ball of aluminum foil and watch where each one stops. They calculate density from mass and volume data using two real rocks and a mystery liquid. They examine reference tables of densities for common solids, liquids, and gases. By the end, they can predict sink vs. float for any substance just by knowing its density relative to water.
8 hands-on stations for teaching how to compare density
A station lab is a student-led activity where small groups rotate through 8 stations (plus a 9th challenge station for early finishers) at their own pace during one to two class periods. You become a facilitator instead of a lecturer. You walk around, spot-check, and break misconceptions while kids work through the rotation.
The Comparing Density Station Lab has four input stations (where students take in new info on mass, volume, density, and buoyancy) and four output stations (where they show what they learned). Here's what's at each one.


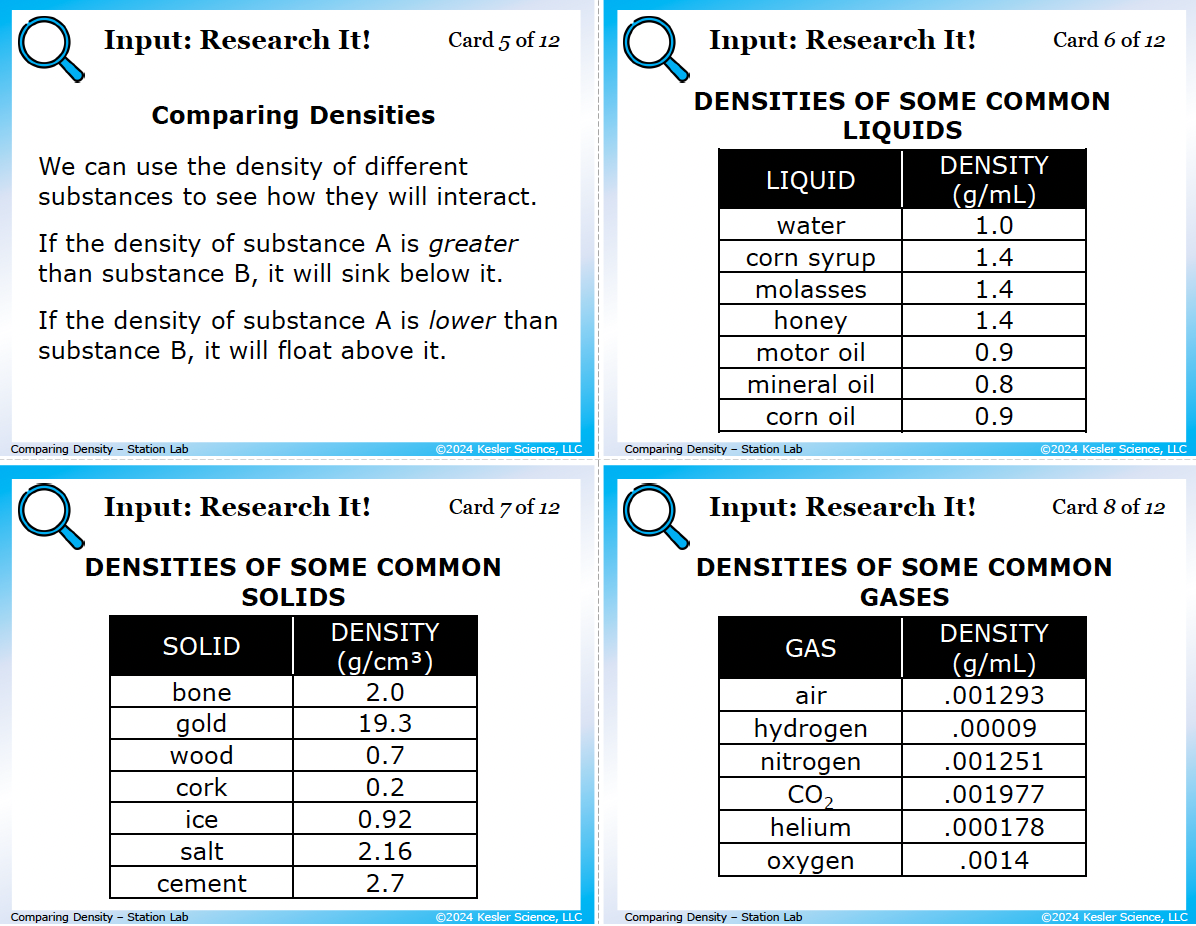
4 input stations: how students learn density
A short YouTube video introduces mass, volume, and the density formula. Kids answer three questions: the difference between mass and matter, the formula for calculating density (D = m/V), and a counterintuitive comparison about volume of two wooden objects of different sizes. Visual learners hook in fast at this station because the video shows the math worked out and the units explained, which makes the formula stick before students see it on a worksheet.
A one-page passage called "How a Lava Lamp Works" explains density through a phenomenon kids actually find cool. The passage walks through the wax blobs (cool wax is denser than the oil so it sinks; the heat lamp warms the wax, the wax expands, becomes less dense, and floats up; cooler wax at the top contracts and sinks again). Vocabulary is bolded throughout (mass, volume, matter, density, buoyancy). Three multiple-choice questions follow, plus the vocab notes section. Comes in two reading levels (Dependent and Modified) plus a Spanish version.
This is the heart of the lab. Kids build a layered density column. They carefully pour 50 mL each of corn syrup (density 1.4 g/mL), water (1.0 g/mL), and vegetable oil (0.9 g/mL) into a clear cup or graduated cylinder, in that order. Then they drop in four solids one at a time: a small ball of aluminum foil, a cork, a coin, and a glass marble. Each one stops at a different layer based on its own density. Three conclusion questions force them to explain which solids are most dense, list the liquids from least to most dense based on what they see, and compare cork vs. water. The visual is unforgettable.
Students examine 8 reference cards: the density formula (D = m/V) with worked example (a bouncy ball with mass 19 g and volume 27 cm³ has density 0.7 g/cm³), units (1 g/cm³ = 1 g/mL), two example rocks with mass and volume given (45 g/15 cm³ vs. 20 g/15 cm³), the rule for comparing densities (denser sinks below, less dense floats above), and three reference tables: densities of common liquids (water 1.0, corn syrup 1.4, motor oil 0.9, etc.), solids (gold 19.3, cork 0.2, ice 0.92), and gases (air 0.001293, hydrogen 0.00009, oxygen 0.0014). Six questions force kids to calculate density from given mass and volume, predict sink-or-float, and explain why ice floats in water.
4 output stations: how students show what they learned
A two-column card sort. Kids sort 9 cards into SINK IN WATER and FLOAT ON WATER columns. Cards include the four solids and one liquid from the Explore It! station (corn syrup, coin, glass marble, cork, vegetable oil, aluminum foil) plus three rule cards (more dense vs. less dense, > 1 g/cm³ vs. < 1 g/cm³, > 1 g/mL vs. < 1 g/mL). Sorting forces kids to connect the experimental result with the actual density value and the qualitative rule. Easy to spot-check at a glance.
Students get a data chart with seven substances and their densities (pine wood 0.4 g/cm³, water 1.0 g/mL, olive oil 0.9 g/mL, gasoline 0.7 g/mL, gold 19.3 g/cm³, wax 0.8 g/cm³, corn syrup 1.4 g/mL) and have to draw what those substances would look like layered in a beaker. Kids have to figure out the order from least dense at top to most dense at bottom, label each layer, and decide where the solids (wood, gold, wax) sink to within the liquid stack. Even kids who say "I can't draw" surprise themselves here because the diagram is just rectangles in the right order.
Three open-ended questions: what would happen if you put oil, water, milk, and honey in a jar, shook it, and let it sit for a day; what happens to a flowing lava lamp when you turn it off and wait a few hours; and how you'd test whether a collection of small solid objects is more or less dense than water. Forces kids to apply density to brand-new scenarios. This is the writing practice middle schoolers need and rarely get in science class.
Eight multiple-choice and fill-in-the-paragraph questions tied to TEKS 6.6D vocabulary (mass, volume, matter, density, buoyancy). Includes what an object less dense than the surrounding liquid does, identifying the densest layer in a four-liquid stack diagram (honey, water, liquid soap, ethyl alcohol), and predicting sink vs. float for a solid with density 2.1 g/cm³ in water (1 g/mL). The fill-in paragraph weaves all five vocabulary words together. If you're grading the lab, this is the easiest station to grade.
Bonus Challenge It! station for early finishers
Four optional extensions: a Sink or Float? investigation where kids predict and test five classroom objects in water, a 10-question density quiz they design for a classmate (with answer key), an acrostic poem using "DENSITY" with illustrations, or watching the classic Diet Coke vs. regular Coke video and explaining (using density) why one floats and the other sinks. Requires teacher approval before they start.
How this fits into a complete comparing density unit
This Station Lab is the Explore day of our full Comparing Density Complete 5E Lesson for TEKS 6.6D. The complete two-week unit follows the 5E method of instruction and includes an Engage hook, the Comparing Density Station Lab for Explore, PowerPoint slides and interactive notebook pages for Explain, student choice projects to Elaborate, and an Evaluate assessment.
Most teachers grab the full 5E because the Station Lab lands hardest with the days around it. But if you just need a strong hands-on day on density of solids and liquids, the Station Lab on its own does the job.
Materials needed to teach comparing density
Materials beyond what's in the download:
- One clear cup, beaker, or 250 mL graduated cylinder per station rotation for the density column.
- 50 mL each of corn syrup, water, and vegetable oil per group. A standard bottle of corn syrup from the grocery store covers a class of 30 with leftovers.
- Four small solids per group: aluminum foil (to roll into a tight ball), a cork, a coin (a penny works), and a glass marble. All cheap, all reusable across all rotations.
- Paper towels for cleanup. Corn syrup is sticky.
- Colored pencils or markers for the Illustrate It! station's layered-beaker drawing.
- Pencils and the printed answer sheets (included)
- A device with internet for the Watch It! station
Standard covered: Texas TEKS 6.6D —
Calculate density to identify an unknown substance. Compare and contrast the densities of solids, liquids, and gases. Supporting Standard.
See the full standard breakdown →Grade level: 6th grade physical science
Time: One to two class periods (45–110 minutes total). Plan for two periods the first time you run a station lab.
Common student misconceptions this lab fixes
- "Bigger things are heavier. So bigger things must be more dense."
This is the big one. Sixth graders confuse mass with density on day one. The Watch It! video question hits it directly with two wood objects of different sizes. The bigger one has more mass, but both have the same density because they're made of the same wood. The Read It! lava lamp passage drives the same point home: the wax doesn't gain or lose mass when it warms up, but its volume increases, so its density drops and it floats. The Explore It! density column then makes it physical. The aluminum foil ball is small but sinks. The cork is similar size but floats. Same volume, very different densities. By the end, kids stop using "big" as a stand-in for "heavy" and "dense."
- "If something is heavy, it sinks. If it's light, it floats."
This is the second-day version of the first misconception. The Explore It! density column catches it. The glass marble is small and pretty light. It still sinks all the way to the bottom because its density is well above 1 g/cm³. A heavy log of pine wood (0.4 g/cm³) floats. What matters isn't weight, it's density relative to the liquid. The Research It! sink-or-float rule (denser sinks below, less dense floats above) makes the rule explicit. The Organize It! card sort then forces kids to put cork (light AND less dense) in the float column and a coin (light but more dense than water) in the sink column. The wording "heavy" disappears once kids commit to the density value as the deciding factor.
- "Density is the same as weight. They mean the same thing."
Many 6th graders use mass, weight, and density interchangeably until they're forced to define them apart. The Research It! density formula card spells out the relationship: D = m/V. Density is the ratio of mass to volume, not just the mass. The Research It! reference tables show two substances with very different masses but the same density (gold has high mass per volume; cork has very low mass per volume). The Assess It! fill-in paragraph forces kids to use mass, volume, density, matter, and buoyancy correctly in one paragraph, and the only way to do that is to keep the words distinct. Once kids see density as a ratio, the confusion fades.
What you get with this comparing density activity
When you buy the Station Lab, you get a single download with everything you need:
- Print version at two reading levels (Dependent for on-grade, Modified for additional support) plus a Spanish Read It! passage
- Digital version as PowerPoint files (works in Google Slides too) at both levels for 1:1 classrooms or Google Classroom
- Teacher Directions and Answer Key for both versions, all keys included
- Station task cards ready to print, laminate, and drop in baskets at each station
- Reference cards for the Research It! station (density formula with worked example, units, two practice rocks, sink-or-float rule, and full density tables for common liquids, solids, and gases)
- Sort cards for the Organize It! station (9 cards: 4 solids, 2 liquids, plus 3 quantitative rule cards for sink/float)
- Substance density data chart for the Illustrate It! station
- Student answer sheets for each level
Tips for teaching comparing density in your 6th grade classroom
Two things make this lab go smoother the first time:
1. Pre-pour the corn syrup and pre-roll the foil.
Corn syrup is the slowest-pouring liquid on the planet, and the first time I ran a similar setup I had groups still measuring out 50 mL when their rotation timer went off. Now I pre-pour 50 mL of corn syrup into each group's cup before class starts. Same with the aluminum foil ball: I pre-roll one tight ball per group so they don't waste five minutes wadding foil. The kids still get the full layered-column experience, the rotation actually finishes on time, and the cleanup is much faster.
2. Use clear cups and plain colored liquids.
The whole point of the density column is that kids can SEE three distinct layers. If your corn syrup is dyed dark brown and your vegetable oil is yellow and your water is clear, the layers pop. If everything is clear, kids squint and can't tell where one stops and another begins. A drop of food coloring in the water (blue or red) makes the boundaries obvious without changing the density meaningfully. Same applies to the solids: a shiny coin and a clear glass marble are easier to spot than two dark objects.
Get this comparing density activity
Or if you want the full two-week experience with the Engage hook, Explain day, Elaborate extension, and Evaluate assessment all included:
(Station Lab is included)
Frequently asked questions
What does TEKS 6.6D cover?
Texas TEKS 6.6D asks 6th grade students to calculate density to identify an unknown substance and to compare and contrast the densities of solids, liquids, and gases. Students should be able to use D = m/V to compute a density value, recognize the units (g/cm³ for solids and g/mL for liquids and gases), and predict whether one substance will sink or float in another based on their relative densities. The standard pairs naturally with 6.6A (particles), 6.6B (mixtures), and 6.6C (classifying elements).
Is this kids' first time with the density formula?
Yes for most 6th graders. They've seen the words "sinks" and "floats" since elementary school, but D = m/V as a formula they have to plug numbers into is brand new. The Watch It! video introduces it, the Research It! reference cards work an example with a bouncy ball, and the Research It! questions force kids to calculate density from given mass and volume. The Assess It! station tests it with a numeric example.
How long does this comparing density activity take?
One to two class periods (45 to 110 minutes total). The Explore It! station's density-column build is the longest piece (especially the slow corn syrup pour), so plan for two periods the first time you run a station lab. Once your class has the rotation routine down, most groups can finish all 8 stations in one period.
Do I need a lot of supplies for this?
Pretty light. A bottle of corn syrup, a bottle of vegetable oil, water from the tap, and four small solids (foil, cork, coin, glass marble) per group. Total cost for a class of 30: about $15 if you're starting from nothing. The Watch It! station also needs a device with internet.
Can I use this in a 1:1 digital classroom?
Yes. The full digital version (PowerPoint or Google Slides) works in 1:1 classrooms and Google Classroom. Students drag digital cards for the Organize It! sort and complete the Illustrate It! beaker drawing on a digital canvas. The Explore It! density-column build is the hardest piece to digitize, but you can substitute video demonstrations or PhET density simulations if you don't have the supplies.
Related resources
- Texas teacher? See the full TEKS 6.6D standard breakdown for misconceptions, phenomena, and engagement ideas.
- Teaching 6.6C first? Check out our Classify Elements Station Lab for TEKS 6.6C, where students sort metals, nonmetals, and metalloids on the periodic table.
- Heading into 6.6E next? See our Evidence of Chemical Changes Station Lab for TEKS 6.6E, where students identify the four signs of a chemical change.


