ATOMS LESSON PLAN – A COMPLETE SCIENCE LESSON USING THE 5E METHOD OF INSTRUCTION
At the end of the atoms lesson plan, students will be able to describe the structure of atoms, including the masses, electrical charges, and locations of protons, neutrons, and electrons. Also, the students will be able to identify that protons determine an element's identity. Each lesson is designed using the 5E method of instruction to ensure maximum comprehension by the students.
The following post will walk you through each of the steps and activities from the atoms lesson plan.
ENGAGEMENT
Objective Introduction
At the beginning of the lesson, the class does a Think-Pair-Share to discuss the objective.
Class Activity
Ask the class (use the PowerPoint to show the questions):
- What is the smallest part of matter?
- What is an atom?
- What are the three main parts of an atom?
- Where are these parts located in an atom?
- What does it mean if an atomic particle has an electrical charge?
Student Activity
Using the Bohr Model Template, including PowerPoint, and three different colors of candy, have students make model of these atoms. Assign a code for protons, neutrons, and electrons:
- Hydrogen
- Helium
- Lithium
- Beryllium
- Boron - (The Moron)
As you work through these atom models, ask students of they start to notice any patterns. Also, introduce atomic mass. Students will have practice during the station lab, so it is okay of they are unsure after this activity.
The teacher will help to clear any misconceptions about atoms. A major misconception is students may think atoms are two-dimensional by using this model.
Estimated Class Time for the Engagement: 20-30 minutes
EXPLORATION
This student-centered station lab is set up so students can begin to explore acids and bases. Four of the stations are considered input stations where students are learning new information about natural selection, and four of the stations are output stations where students will be demonstrating their mastery of the input stations. Each of the stations is differentiated to challenge students using a different learning style. You can read more about how I set up the station labs here.
EXPLORE IT!
Students will be working in pairs to create atom models using a Bohr template. Students will have to follow the directions on the task cards and using the small squares that represent the subatomic particles (protons, neutrons, and electrons) create certain atoms. Cards will also inform students about the charges of each subatomic particle.

WATCH IT!
At this station, students will be watching a video describing atoms. Students will then answer some questions relating to the video and record their answers on their lab station sheet. For example, What is an atom? What is at the center of the atom? What two subatomic particles make up the center of the atom? What kind of charge do protons, neutrons, and electrons have?
RESEARCH IT!
The research station will allow students to interact with virtual atom creator using the PhET website. Students will follow the directions on the task cards to create atoms based on the number of protons, neutrons, and electrons listed. Students will have to build and identify the atom. As students build a random atom, they will discover which subatomic particle determines the identity of the atom.
READ IT!
This station will provide students with a one page reading about atomic structure. In the reading, students will discover how the atom is the smallest unit of matter. They will learn about the basic structure of the atom. Students will learn about the charges of each of the parts, protons, neutrons, electrons. There are 4 follow-up questions that the students will answer to show reading comprehension of the subject.
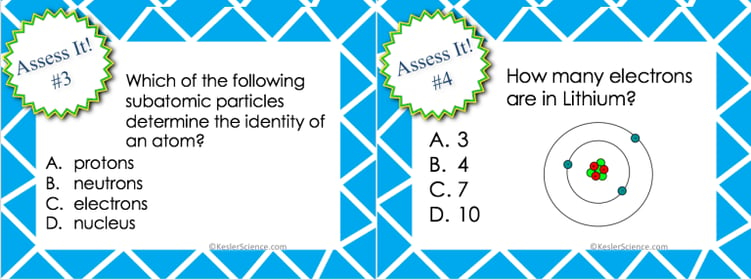
ASSESS IT!
The assess is station is where students will go to prove mastery over the concepts they learned in the lab. The questions are set up in a standardized format with multiple choice answers. Some questions will ask students: Identify the correct statement from 4 four choices that talk about atoms. What is the role of electrons in an atom? Which of the following subatomic particles determine the identity of an atom? How many electrons are in Lithium?
WRITE IT!
Students who can answer open-ended questions about the lab truly understand the concepts that are being taught. At this station, the students will be answering three task cards to explain the differences between protons, neutrons, and electrons. Describe the nucleus of the atom. Which subatomic particle determines the identity of the atom?
ILLUSTRATE IT!
Your visual students will love this station. Students will recreate an Oxygen atom on their lab sheet. Students will then have to label everything about their model.

ORGANIZE IT!
The organize it station allows your students to place cards in the correct column and row that describes the location, charge, and size of protons, neutrons, and electrons.
Estimated Class Time for the Exploration: 1-2, 45 minute class periods
EXPLANATION
The explanation activities will become much more engaging for the class once they have completed the exploration station lab. During the explanation piece, the teacher will be clearing up any misconceptions about atoms with an interactive PowerPoint, anchor charts, and interactive notebook activities.
The atoms lesson includes a PowerPoint with activities scattered throughout to keep the students engaged.
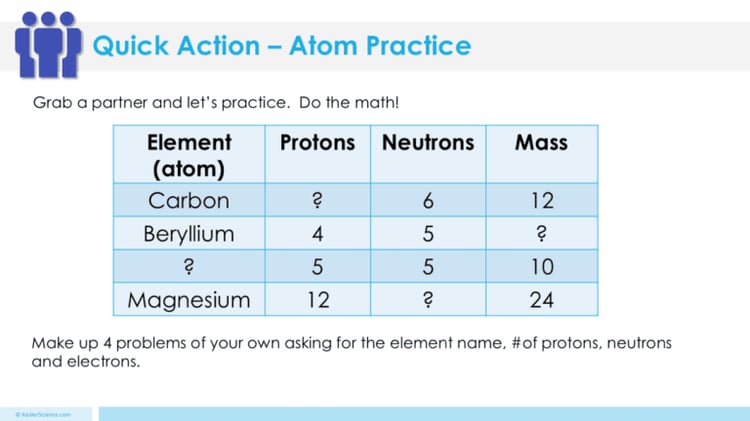
The students will also be interacting with their journals using INB templates for atoms. Each INB activity is designed to help students compartmentalize information for greater understanding of the concept. The atoms INB templates allow students to focus their notes on identifying the descriptions and charges of each subatomic particle. Also, the INB templates will help students to understand the information provided on a chemical symbol, and how to determine the atomic mass and number of protons and electrons from a symbol using the APE-MAN acronyms.

Estimated Class Time for the Exploration: 2-3, 45 minute class periods
ELABORATION
The elaboration section of the 5E method of instruction is intended to give students choice on how they can prove mastery of the concept. When students are given choice the ‘buy-in’ is much greater than when the teacher tells them the project they will have to create. The elaboration project will allow students to create a presentation to teach about atoms.
Estimated Class Time for the Elaboration: 2-3, 45 minute class periods (can also be used as an at-home project)
EVALUATION
The final piece of the 5E model is to evaluate student comprehension. Included in every 5E lesson is a homework assignment, assessment, and modified assessment. Research has shown that homework needs to be meaningful and applicable to real-world activities in order to be effective. When possible, I like to give open-ended assessments to truly gauge the student’s comprehension.
Estimated Class Time for the Evaluation: 1, 45 minute class period
DOWNLOAD THE FULL LESSON NOW
The full lesson is available for download from the Kesler Science Store. Save yourself a ton of time and grab it now.
Download Over $100 in FREE Resources
For Middle School Science
Simply create a login below and gain immediate access to a selection of our Kesler Science product line worth $100 - for FREE. There's a full version of every product type! You'll also join tens of thousands of middle school science teachers who receive timely tips and strategies straight to their inbox.






