Conservation of Mass Activity: 8 Hands-On Stations for Teaching the Law of Conservation of Mass and Balanced Equations (TEKS 8.6E)
If you've ever lit a candle in front of an 8th grader and asked, "where did the wax go?", you already know the answer they're going to give. "It disappeared." Or they'll say it "burned up." Both wrong, both wildly common.
The law of conservation of mass is one of those topics where the vocabulary is easy and the actual idea is hard. Atoms don't disappear in chemical reactions. They rearrange. Mass on the reactant side has to equal mass on the product side. Always.
The Conservation in Reactions Station Lab for TEKS 8.6E walks kids through this in one to two class periods. They weigh a glow stick before and after they crack it. They drop an Alka-Seltzer tablet in a sealed bag and watch the mass stay the same while the bag puffs up. By the end, they're not just reciting "matter can't be created or destroyed." They've watched it.
8 hands-on stations for teaching conservation of mass
A station lab is a student-led activity where small groups rotate through 8 stations (plus a 9th challenge station for early finishers) at their own pace during one to two class periods. You become a facilitator instead of a lecturer. While kids work, you walk around, spot-check, fix misconceptions on the fly, and watch for the moment things click.
The Conservation in Reactions Station Lab has four input stations (where students take in new info on conservation of mass and balancing equations) and four output stations (where they show what they learned). Here's what's at each one.

4 input stations: how students learn conservation of mass
A short YouTube video introduces the law of conservation of matter. Students answer three questions: what must be true during any chemical reaction, how the law applies to a copper pipe rusting, and what happens to the mass of water when it boils away as steam. (Spoiler: the steam still has mass. Most kids miss this the first time.) Visual learners get traction here.
A one-page passage called "Chemistry's Balancing Act" walks through reactants, products, and the law of conservation of mass. Photosynthesis is the worked example, since TEKS 8.6E specifically calls it out. Three reading-comprehension questions follow. The passage comes in two reading levels (Dependent and Modified) plus a Spanish version, so you can differentiate without prepping anything.
This is the heart of the lab. Students do two hands-on experiments with a digital scale. First, they weigh a glow stick before and after cracking it. The mass stays the same. Then they put an Alka-Seltzer tablet and 10 mL of water in a sealed Ziploc bag, weigh it, drop the tablet in, seal the bag fast, and weigh it again. Bag puffs up with CO₂, mass stays the same. They've just proven conservation of mass with their hands. The questions that follow ask why there's any tiny mass difference (escaping gas, scale precision) and what they'd predict for an open container.
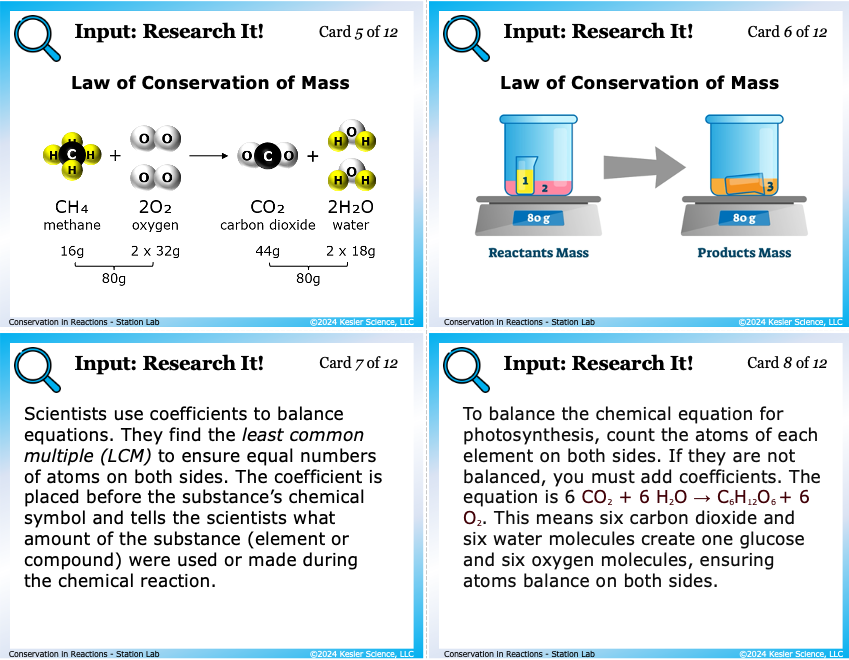
Students examine 12 reference cards showing chemical equations as both molecule diagrams and balance-scale visuals. They start with the unbalanced N₂ + H₂ → NH₃, then see the balanced N₂ + 3H₂ → 2NH₃. They count atoms on each side, define reactants and products, and figure out exactly what coefficients are doing. This is where the Texas TEKS 8.6E phrase "rearrangement of atoms using chemical equations" stops being abstract.

4 output stations: how students show what they learned
A card sort. Kids physically arrange chemical equations under two headers: Balanced and Unbalanced. Equations include 2 Al + 3 H₂SO₄ → Al₂(SO₄)₃ + 3 H₂, 4 Fe + 3 O₂ → 2 Fe₂O₃, and Fe₂O₃ + C → 2 Fe + CO₂ (unbalanced). Easy to spot-check at a glance, and the fastest way to see who's actually counting atoms vs. who's just guessing.
Students draw a model of H₂ + Cl₂ → HCl, color-code the elements, label reactants and products, balance the equation, and rewrite it with coefficients. This station catches the biggest misconception in the standard: that you can change subscripts to balance an equation. (You can't. They define what the substance is.) Even kids who say "I can't draw" get this one.
Three open-ended questions where students explain in their own words why reactant mass equals product mass, how conservation of mass connects to balancing equations, and a real-world example. This is the writing practice middle schoolers need and rarely get in science class.
Eight multiple-choice and fill-in-the-paragraph questions tied to TEKS 8.6E vocabulary (atoms, chemical reaction, law of conservation of mass, products, reactants). Includes the photosynthesis equation, an iron-and-sulfur calculation, and a check on which equation actually demonstrates conservation. If you're grading the lab, this is the easiest station to grade.
Bonus Challenge It! station for early finishers
Four optional extensions: a "What If?" stoichiometry problem with methane combustion, a balancing-equations game students design themselves, an infographic, and a research task on the history of the law of conservation of mass. Requires teacher approval before they start.
How this fits into a complete conservation of mass unit
This Station Lab is the Explore day of our full Conservation in Reactions Complete 5E Lesson for TEKS 8.6E. The complete two-week unit follows the 5E method of instruction and includes an Engage hook, the Conservation in Reactions Station Lab for Explore, PowerPoint slides and interactive notebook pages for Explain, student choice projects to Elaborate, and an Evaluate assessment that wraps the whole unit.
Most teachers grab the full 5E because the Station Lab lands hardest with the days around it. But if you just need a strong hands-on day on conservation of mass and balancing equations, the Station Lab on its own does the job.
Materials needed to teach conservation of mass with glow sticks and Alka-Seltzer
Materials beyond what's in the download:
- Digital scale that measures in grams, ideally with a tare button. The cheap kitchen scales work fine. One per Explore It! station basket is plenty.
- Glow sticks (one per group rotation, or one per student if you want them to keep them). Dollar store glow sticks are perfect.
- Alka-Seltzer tablets (one per group rotation). A box of generic store-brand antacid tablets is cheaper and works the same.
- Ziploc snack-size bags (one per group rotation). Sealable is the whole point. The gas needs to stay in.
- Water and a way to measure 10 mL (small graduated cylinder or a plastic medicine cup).
- Colored pencils or markers for the Illustrate It! station.
- Pencils and the printed answer sheets (included)
- A device with internet for the Watch It! station
Standard covered: Texas TEKS 8.6E —
Investigate how mass is conserved in chemical reactions and relate conservation of mass to the rearrangement of atoms using chemical equations, including photosynthesis. Readiness Standard.
See the full standard breakdown →Grade level: 8th grade chemistry (works as a stretch lesson for advanced 7th)
Time: One to two class periods (45–110 minutes total). Plan for two periods the first time you run a station lab.
Common student misconceptions this lab fixes
- "When something burns, the mass disappears."
Nope. The atoms combine with oxygen and float away as gases (mostly CO₂ and water vapor). If you could capture all those gases on a scale, the mass would balance. The Alka-Seltzer-in-a-sealed-bag activity is built specifically to surface this. Mass stays put when the gas is trapped. Open the bag, and now the kids see why combustion looks like "disappearing" matter when it isn't.
- "You can change subscripts to balance an equation."
This is the single biggest error in this standard. Subscripts define what a substance IS — H₂O has two hydrogens because it's water. Change the subscript and you've got a different substance. Coefficients are the only thing you can change to balance. The Illustrate It! station catches this directly. If a kid's drawing turns H₂ into H₃ to make the equation work, you know to slow down at the Explain day.
- "Gases don't really have mass."
They do. Air has mass. CO₂ has mass. Steam has mass. The trick is that gases drift up and out of an open container, so it looks like the mass left. The glow stick activity shows mass conservation in a sealed system. The standard's photosynthesis example is another good one — kids forget to count the gases (CO₂ in, O₂ out) when they're calculating mass. The Read It! passage hits this head-on.
What you get with this conservation of mass activity
When you buy the Station Lab, you get a single download with everything you need:
- Print version at two reading levels (Dependent for on-grade, Modified for additional support) plus a Spanish Read It! passage
- Digital version as PowerPoint files (works in Google Slides too) at both levels — for 1:1 classrooms or Google Classroom
- Teacher Directions and Answer Key for both versions, all keys included
- Station task cards ready to print, laminate, and drop in baskets at each station
- Reference cards for the Research It! station (the chemical equation and balance-scale diagrams)
- Sort cards for the Organize It! station (balanced and unbalanced equations)
- Student answer sheets for each level
No login required. Download once, use forever. Reprint as many times as you want.
Tips for teaching conservation of mass in your 8th grade classroom
Two things make this lab go smoother the first time:
1. Pre-stage the Alka-Seltzer setup.
Have the Ziploc bags pre-measured with 10 mL of water sitting at the Explore It! station, with a dry Alka-Seltzer tablet next to each one in a small cup. Speed matters. The whole point is to seal the bag fast enough that no CO₂ escapes. If kids spend three minutes hunting for a baggie and another two pouring water, the science gets lost. Pre-stage and the activity takes about four minutes per group.
2. Stand near Explore It! during the first rotation.
Watch the moment kids put the bag back on the scale and see the same mass as before. Some will say "the scale is broken." That's the moment. Lean in and ask, "Where did the gas go?" When they realize the gas is still in the bag, conservation of mass clicks for life. If you don't see that moment with the first group, slow the pacing for the rest of the class.
Get this conservation of mass activity
Or if you want the full two-week experience with the Engage hook, Explain day, Elaborate extension, and Evaluate assessment all included:
(Station Lab is included)
Frequently asked questions
What does TEKS 8.6E cover?
Texas TEKS 8.6E asks 8th grade students to investigate how mass is conserved in chemical reactions and relate conservation of mass to the rearrangement of atoms using chemical equations, including photosynthesis. It's a Readiness Standard, which means it shows up on STAAR with weight. Students should be able to count atoms on each side of an equation, balance equations using coefficients, and explain that atoms in a reaction are rearranged, not created or destroyed.
What's the difference between a coefficient and a subscript?
A subscript is the small number after an element symbol that tells you how many of that atom are in the molecule (the 2 in H₂O). It defines what the substance is. A coefficient is the big number in front of a chemical formula that tells you how many molecules of that substance are in the reaction (the 6 in 6 H₂O). To balance an equation, you only change coefficients. Changing a subscript changes the substance itself.
How long does this conservation of mass activity take?
One to two class periods (45 to 110 minutes total). The first time you run a station lab, plan for two periods so kids can learn the rotation and pacing without feeling rushed. Once your class has the routine down, most groups can finish all 8 stations in one period.
Do I need to provide my own materials?
Yes, but the materials are cheap and easy. You'll need a digital scale, glow sticks, Alka-Seltzer or generic antacid tablets, snack-size Ziploc bags, water, and colored pencils. Total cost for a class of 30: about $20 if you already own a digital scale. The Watch It! station also needs a device with internet.
Can I use this for 7th grade or in a 1:1 digital classroom?
Yes to both. The Modified version of every station works as a stretch lesson for advanced 7th graders. The full digital version (PowerPoint or Google Slides) works in 1:1 classrooms and Google Classroom. Students type into the slides and submit them back. The Explore It! station still needs the physical scale and Alka-Seltzer for the full experience, but you can substitute a video demo for fully remote settings.
Related resources
- Texas teacher? See the full TEKS 8.6E standard breakdown for misconceptions, phenomena, and engagement ideas.
- Going deeper into chemical reactions? Our complete 5E balancing chemical equations lesson walks through the full unit.


